Nitrogen narcosis
Background
- Toxic effects of breathing nitrogen-containing gases while at depth
- Called the "rapture of the deep"
- Develops between 3-4ATM (99-132 feet)[1]
- Can be prevented by using nitrogen-free gas mixture (e.g. heliox), or mixtures with reduced nitrogen content (e.g. helium-nitrogen-oxygen)[1]
- Risk factors = exertion during dive, cold conditions, alcohol intoxication before dive[1]
- Other inert gases cause similar symptoms at depth - severity based on lipid solubility of the gas[3]
Diving Physiology
- Pascals Law applies to the diving body (without air filled areas such as lungs) states that the pressure applied to any part of the enclosed liquid will be transmitted equally in all directions through the liquid.
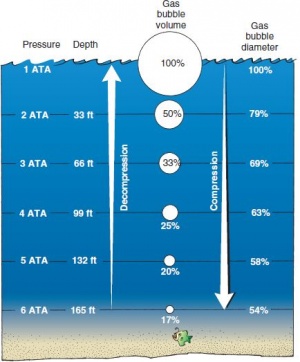
- Boyles Law applies to the diving body's air filled areas such as lungs, sinuses, middle ear, and states that the volume and pressure of a gas at a given temperature are inversely related.
- At 2 ATA (10m/33ft) a given gas would be 1/2 it's volume, at 3 ATA (20m/66ft) it would be 1/3 it's volume and so on.
- Dalton's Law applies to the total pressure of an ideal gas mixture being the sum of the partial pressures of each individual gas.
- Divers may used Enriched Air NITROX mixtures to proportionally increase partial pressures of oxygen and reduce partial pressures of nitrogen while diving.
- At extremes of depth, additional inert gasses such as helium in TRIMIX are used to further reduce partial pressures of both oxygen and nitrogen below toxic levels.
- Henry's Law applies to the dissolvability of gasses into fluids, including body tissues, being proportional to the partial pressure of the gas.
- The increased pressure at depth causes divers to breath their gas mix at increased pressure to defeat the external water pressure.
- Increased inhaled partial pressures of nitrogen increase risk of nitrogen narcosis, and dissolved nitrogen in tissues re-expanding in micro-bubbles on ascent is the essential cause of decompression sickness. This can affect divers at any depth, including commonly-seen recreational diving depths of 20m/60ft or less.
- Increased inhaled partial pressures of oxygen, generally beyond 1.4-1.6atm, increases risk of oxygen toxicity. This is typically not a substantial risk in common depths of recreational divers at 20m/60ft of depth or less, but can be for more advanced divers at deeper depths.
- The increased pressure at depth causes divers to breath their gas mix at increased pressure to defeat the external water pressure.
Clinical Features
- Insidious onset of symptoms similar to alcohol intoxication (or nitrous oxide inhalation)[1][2]
- Early stage: Euphoria, false sense of security, impaired judgement, diminished reaction times
- Later stage: Impaired concentration and memory, paresthesias, hallucinations
- Final stage: [Occurs at depths of at least 10-13ATM (333-429 feet)], lethargy and loss of consciousness
- Causes high risk of making bad decisions at depth, i.e. impairs diver's judgment therefore leading to drowning accidents (cause of up to 9% of diving deaths)[1][2]
Differential Diagnosis
Diving Emergencies
- Barotrauma of descent
- Otic barotrauma
- Pulmonary barotrauma
- Sinus barotrauma
- Mask squeeze
- Barodentalgia (trapped dental air causing squeeze)
- Barotrauma of ascent
- Pulmonary barotrauma (pulmonary overpressurization syndrome)
- Decompression sickness (DCS)
- Arterial gas embolism
- Alternobaric vertigo
- Facial baroparesis (Bells Palsy)
- At depth injuries
- Oxygen toxicity
- Nitrogen narcosis
- Hypothermia
- Contaminated gas mixture (e.g. CO toxicity)
- Caustic cocktail from rebreathing circuit
Evaluation
- Clinical diagnosis
Management
- Ascent - symptoms completely reversed within minutes by ascending to a shallower depth, with no long-term effect
- Therefore, consider other causes in patients with continued symptoms after ascent
Disposition
- Discharge
See Also
External Links
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Grover CA, Grover DH. Albert Behnke: nitrogen narcosis. J Emerg Med. 2014 Feb;46(2):225-7.
- ↑ 2.0 2.1 2.2 Levett DZ, Millar IL. Bubble trouble: a review of diving physiology and disease. Postgrad Med J. 2008 Nov;84(997):571-8.
- ↑ Bove AA. Diving medicine. Am J Respir Crit Care Med. 2014 Jun 15;189(12):1479-86.