COVID-19
See also prevention of COVID-19 transmission in the healthcare setting and COVID-19 in pregnancy
Background
- The current national and international pandemic is from a virus named SARS-CoV-2 (previously 2019-nCoV), which causes a disease named COVID-19 (also known as "2019 Novel Coronavirus")
- First detected in Wuhan, China
Clinical Features
Initial Presentation
- At onset: fever, dry cough, myalgias, fatigue, shortness of breath
- Fever and cough start early, shortness of breath noted about 9 days into illness
- Fever not present in all adults (less common in vulnerable populations)
- Less common: cough with sputum, sore throat, headache, congestion, GI symptoms
Risk Factors for Severe Disease
- Older adults, underlying conditions (lung disease, heart disease, diabetes)
- Children: milder disease
- Pregnant patients: don’t appear to be at increased risk of infection or adverse outcomes (limited data - see COVID-19 in pregnancy)
Common Complications
- Most common complications: pneumonia, ARDS (avg 8 days from onset, 20% of patients in China)
- Decompensation risk occurs during 2nd week of illness leading to respiratory failure
Differential Diagnosis
Influenza-Like Illness
- Influenza
- Parainfluenza
- URI
- Pneumonia
- Sinusitis
- Toxic exposure
- Pyelonephritis
- Bronchitis
- Coronavirus
Causes of Pneumonia
Bacteria
Viral
- Common
- Influenza
- Respiratory syncytial virus
- Parainfluenza
- Rarer
- Adenovirus
- Metapneumovirus
- Severe acute respiratory syndrome (SARS)
- Middle east respiratory syndrome coronavirus (MERS)
- 2019-nCoV (COVID-19)
- Cause other diseases, but sometimes cause pneumonia
Fungal
- Histoplasmosis
- Coccidioidomycosis
- Blastomycosis
- Pneumocystis jirovecii pneumonia (PCP)
- Sporotrichosis
- Cryptococcosis
- Aspergillosis
- Candidiasis
Parasitic
Evaluation
Workup
- LA County DPH checklist (http://publichealth.lacounty.gov/acd/ncorona2019/checklist.htm)
- Mild illness: DO NOT send for testing (increased risk of exposure to COVID-19)
- ER: DO NOT go unless hospital level of care is needed (increased exposure of other patients and staff)
- Testing can be done in ambulatory setting if absolutely needed (see precautions)
Viral Test
- In the United States, the US Centers for Disease Control is distributing the CDC 2019-Novel Coronavirus (2019-nCoV) Real-Time RT-PCR Diagnostic Panel to public health labs through its International Reagent Resource[1]
- Internationally the WHO has distributed kits based on the RT-PCR platform[2]
- BIOFIRE RESP PANEL Corona Virus assay does NOT detect this subtype - DO NOT USE
Broad Testing Recommendations
The CDC has expanded testing beyond the initial travel to china recommendations[3]
- Test any person who has fever or respiratory illness AND had close contact with a known COVID-19 patient
- Test any person who has fever or respiratory illness AND a history of travel to CDC-classified high risk areas or areas with recent significant community spread
- Test any person who has severe respiratory illness AND is being admitted to the hospital
- Test any hospitalized person who has respiratory illness AND no alternative diagnosis
- Consider testing any person who has respiratory illness AND lives in a residential facility such as a nursing home or recently returned from a cruise
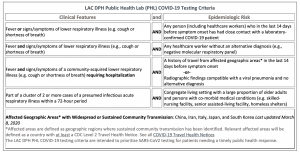
Guidelines: Epidemiologic Factors
- Persons (including HCW) within 14 days of travel (domestic/international), or
- Close contacts with lab-confirmed COVID19 patient within 14 days
Criteria For Sending Specimen to PHL
Criteria For Sending Specimen to Commercial Lab
- Patients with fever and cough/shortness of breath not requiring hospitalisation who have:
- History of travel from affected geographic areas (domestic / international) within 14 days of symptom onset
- Other exposure risk indicated by the patient’s history and clinical judgement (and no alternative diagnosis -- e.g. negative flu test)
Decision To Obtain Imaging
- CXR: no significant issues with contamination/disinfection
- CT: Temporarily out of commission after COVID19 patient in scanner
Clinical Sample Collection
- Best way to collect:
- Upper respiratory tract and lower tract specimens (if available).
- NP swabs
- Put both of them in the same tube and send for a single test
- For productive cough patients: can collect sputum to send for testing. CDC does NOT recommend inducing sputum (because aerosol generating)
- Upper respiratory tract and lower tract specimens (if available).
Testing: Turnaround Time
- LA County Public Health Lab (PHL) = ~ 2 business days
- Commercial lab = ~ 3-4 business days
Diagnostic Findings
- Lymphopenia most common in critically ill; mildly elevated ALT, AST; normal pro-calcitonin on admission
- Elevated d-dimer and severe lymphopenia are associated with increased mortality
- RT-PCR is currently test of choice for confirming cases
- Test kit availability is currently limited as of mid March
- Consider influenza/viral respiratory panel to identify alternative diagnoses
- Although co-infection is possible
Imaging
Xray
- Portable CXR preferred in PUI to prevent spread of infection
- May be normal in early disease
- Typical pattern is peripheral patchy ground glass opacities (GGO)
- More opacities correlates with worse disease
- GGOs may coalesce and appear as infiltrates
CT
- Many have normal imaging early on (so CDC DOES not recommend CT for diagnostic purposes at this time)
- CT (86%) more sensitive than CXR (59%) for detecting GGOs
- Radiopaedia COVID-19 Resources (https://radiopaedia.org/articles/covid-19)
- From the American College of Radiology (3/11/20): “Generally, the findings on chest imaging in COVID-19 are not specific, and overlap with other infections, including influenza, H1N1, SARS and MERS. Being in the midst of the current flu season with a much higher prevalence of influenza in the U.S. than COVID-19, further limits the specificity of CT.”
- Reinfection (after recovery from COVID19): unclear if possible
- Limited data. Unlikely to be reinfected shortly after but unknown about later on
Management
See prevention of COVID-19 transmission in the healthcare setting for PPE recommendations
General
- No specific treatment currently available
- Non-pharmaceutical interventions will be most important
- Spread Prevention
- Mitigation strategies
Mild Cases
- Supportive care is mainstay of therapy for patients with mild viral symptoms
- Most patients will do well enough for discharge home
- Discuss with Dept of Public Health, who will guide testing and, if discharging, help patient remain in isolation at home
Respiratory failure
- NIPPV may increase the spread of viral particles via droplets making early intubation the preferred airway management strategy in patients with respiratory distress/failure
- Using 2 viral filters attached to a 2 tube NIPPV circuit in a negative pressure room may sufficiently prevent viral spread
- During BVM, if needed, use a viral filter
- Due to the viral spread risk early intubation is the preferred means of airway managment
Intubation
- High risk procedure for aeresolization
- Patient ideally in negative pressure room. Limit individuals in room to essential staff only.
- PPE for all in room: N95, gown, gloves, eye shield
- Minimum PPE for provider intubating: same as above (N95, gown, gloves, eye shield)
- Optional PPE for provider intubating: PAPR, double glove, double gown, shoe covers, buddy system for donning/doffing
- Use BVM with viral filter
- Use sufficient paralytics to prevent coughing gagging
- Most experienced provider should perform intubation.
- Ventilate using ARDSnet protocol
Investigational Agents
Generally not started in ED setting
- Remdesivir (IV)
- CDC does not recommend for or against any investigational therapies at this time
- Contact Gilead directly for use: compassionateaccess@gilead.com
- Background: novel antiviral nucleotide analog. Initially developed for Ebola and Marburg (has since been found to show activity against other single stranded RNA viruses such as RSV, Lassa fever virus, Nipah virus and the coronaviruses including MERS and SARS)
- 3 clinical trials across country (one is NIH adaptive trial)
- 2 other trials are investigational open-label trials testing different dosages for moderate or severely hospitalized patients
- Ritonavir also being used but no data available. Same for chloroquine.
Contraindicated
- High-dose corticosteroids should be AVOIDED (due to progression of viral replication reported from prior coronaviruses; e.g. MERS, SARS)
- Avoid nebulizers as they are generally ineffective may aerosolize virus
- Albuterol with spacer is safer, though probably ineffective unless co-occuring reactive airway disease
- Generally avoid BiPAP and high-flow nasal oxygen as these may increase viral spread
- WHO cautiously states that high flow oxygen may be occasionally indicated.
- There is anecdotal concern about NSAID use; some have suggested preferentially using acetaminophen however, there are no formal recommendations to avoid NSAIDs at this time
Disposition
- Mild cases for persons under investigation for Covid-19 awaiting a positive test result can self quarantine at home in conjunction with the local Public Health Dept
- If admitting, needs to be placed in negative pressure isolation room with airborne and droplet precautions
Decision To Hospitalize
- Mild symptoms may go home and self-isolate/quarantine
- Note: symptoms may worsen over 2nd week of illness
- Hospitalize: Respiratory distress/failure, multi-organ failure, rapid disease progression requiring escalating supportive care
- May consider discontinuation of hospital isolation when:
- Resolution of fever without anti-pyretic, resolution of symptoms, and negative COVID19 testing
See Also
External Links
Video
{{#widget:YouTube|id=exV5hEG62CY}}
References
- ↑ https://www.internationalreagentresource.org/
- ↑ Sheridan, Cormac . "Coronavirus and the race to distribute reliable diagnostics". Nature Biotechnology https://www.nature.com/articles/d41587-020-00002-2
- ↑ Criteria to Guide Evaluation and Laboratory Testing for COVID-19 https://www.cdc.gov/coronavirus/2019-nCoV/hcp/clinical-criteria.html