Tetanus prophylaxis
(Redirected from Tetanus vaccine)
Background
- Tetanus immunoglobulin (TIG) and the tetanus vaccine (Td) are both crucial in preventing symptomatic tetanus infection
- Incubation period for a tetanus infection: 1 day - 2 months
- 70% of wounds are prone to tetanus. These include contaminated wounds or puncture wounds but 30% are clean wounds. There are also case reports of surgically created wounds with autoclaved instruments resulting in tetanus [1]
Prophylaxis
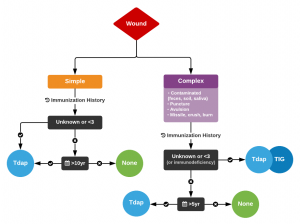
An Algorithm for Tetanus Prophylaxis in Adults [2]
- Past vac unknown or <3 total
- Clean wound → Td
- Dirty wound → Td & TIG
- Past vac 3 or more total
- Clean wound → Td Q10yrs
- Dirty wound → Td Q5yrs
Tetanus Vaccine Selection Guide
| Age | If No Pertussis Contraindications | If Pertussis Contraindicated | Comments |
| 2mo - 7yrs | DTaP | DT | DT available from pharmacy |
| 7yrs - 10yrs | Td | Td | |
| 10yrs | Tdap(Adacel)^ | Td | ^Off label use, but appears safe and immunogenic |
| 11yrs - 16yrs | Tdap(Adacel) | Td | |
| 16yrs - 65 | Tdap(Adacel)^^ | Td | ^^Use Td if patient is known to have received prior Tdap |
| 65yrs + | Td |
Td |
Consider TIG for tetanus prone wound plus Td(tetanus immune globulin) |
Caveats
- Adolescents who have already gotten a booster dose of Td are encouraged to get a dose of Tdap as well. Waiting 5 years after their last Td is encouraged but not required. A period of at least 2 years between Td and Tdap is recommended in these cases.
- Pregnancy is not a contraindication to Tdap. The only true contraindication to tetanus immunization is a history of neurologic or severe hypersensitivity to a previous dose.
- Stable neurologic disorders and family history of vaccine reactions are not contraindications to receiving these vaccines.
- No vaccine has ever been shown to play any role in the development of autism or autism spectrum disorders.
See Also
- Tdap (Pertussis Vaccine) for new recommendations from CDC
- Tetanus (Acute)
- Vaccination schedule


