ST-segment elevation myocardial infarction
Background
- Abbreviation: STEMI
- RV infarction accompanies ~25% of inferior STEMIs
- Hemodynamically significant only 10% of the time
- Do NOT reduce preload (caution with NTG)
- Optimise preload (ensure volume replete)
- Posterior (aka inferolateral) infarction is rarely isolated (~3-8% of all AMIs)
- Treat as STEMI
- Look for reciprocal changes, except in aVR and V1
- Apply V7, V8, V9 leads and repeat ECG looking for ST elevation
- Usually will see changes in V6 OR II, III, aVF
Clinical ACS Categories
- ST-segment elevation myocardial infarction (30%)
- Non-ST-elevation myocardial infarction (25%)
- Unstable Angina (UA) (38%)
- The new title, “Non-ST-Elevation Acute Coronary Syndromes,” emphasizes the continuum between UA and NSTEMI[1]
- NSTEMI myocardium is damaged enough to increase biomarkers, UA is not.
Fourth Universal Definition of STEMI
Any of the following:[2]
- 1 mm of ST elevation in any two contiguous leads except V2 and V3.
- The acceptable degree of ST elevation in V2 and V3 changes based on age and gender.
- In women: 1.5mm elevation in V2 and V3
- In men under 40: 2.5mm elevation in V2 and V3
- In men 40 and older: 2mm elevation in V2 and V3
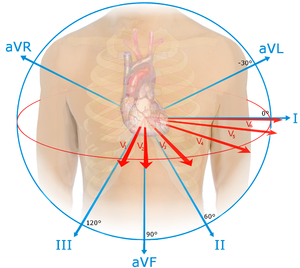
ACS Anatomical Correlation Chart
| Ischemic Changes | Location | Coronary Artery |
| STE V1-V3, TWI Q waves in V1-V3 over time |
Septal | Septal branch |
| STE V2-V4 | Anterior | LAD |
| STE I, aVL, V5, V6 STD inf leads |
Lateral | Circumflex |
| STE I, aVL, V2-6 | Anterolateral | LAD + circumflex = Left main or 2 critical lesions |
| STE II, III, aVF STD in aVL (most common lead to see reciprocal change) |
Inferior | RCA |
|
STE V1 (only lead looking at RV)
|
Right ventricle | RCA |
|
STD in V1, V2, V3; |
Posterior aka Inferolateral | RCA (90%), LCA (10%) |
| STE avR>V1 Doesn't apply in SVT |
Anterolateral | Left Main |
Prehospital
- Hyperoxia may increase myocardial injury
- Avoid supplemental oxygen unless hypoxic[3]
- Activate cath lab for patients with STEMI on prehospital ECG even if ST elevation has resolved by time of arrival at hospital [4]
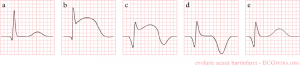
STEMI Stages of Development
| Stage | Duration | Timing | Finding | ECG |
| 1 | 30min - hours | Hyperacute T waves
|
Normalizes in days, weeks, or months | |
| 2 | Minutes - hours | ST segment elevation
|
ST segment resolution occurs over 72hrs; completely resolves within 2-3wks | |
| 3 | Within 1hr; completed within 8-12hr | Q waves | Persist indefinitely in 70% of cases |
Clinical Features
Risk of Acute Coronary Syndrome
Clinical factors that increase likelihood of ACS/AMI:[5][6]
- Chest pain radiating to both arms > R arm > L arm
- Chest pain associated with diaphoresis
- Chest pain associated with nausea OR vomiting
- Chest pain with exertion that is improved with rest
Clinical factors that decrease likelihood of ACS/AMI:[7]
- Pleuritic chest pain
- Positional chest pain
- Sharp, stabbing chest pain
- Chest pain reproducible with palpation
Gender differences in ACS
- Women with ACS:
- Less likely to be treated with guideline-directed medical therapies[8]
- Less likely to undergo cardiac catheterization[8]
- Less likely to receive timely reperfusion therapy[8]
- More likely to report fatigue, dyspnea, indigestion, nausea or vomiting, palpitations, or weakness,[8] although some studies have found fewer differences in presentation[9]
- More likely to delay presentation[8]
- Men with ACS:
- More likely to report central chest pain
Factors associated with delayed presentation[8]
- Female sex
- Older age
- Black or Hispanic race
- Low educational achievement
- Low socioeconomic status
Additional features
Signs/symptoms may include
- Chest pain
- SOB
- Dizziness/lightheadedness, syncope/presyncope
- Nausea/vomiting
- Diaphoresis
- Anxiety, sense of impending doom
- Palpitations
- Dysrhythmias
- Cardiac arrest
Differential Diagnosis
ST Elevation
- Cardiac
- ST-segment elevation myocardial infarction (STEMI)
- Post-MI (ventricular aneurysm pattern)
- Previous MI with recurrent ischemia in same area
- Wellens' syndrome
- Coronary artery vasospasm (eg, Prinzmetal's angina)
- Coronary artery dissection
- Pericarditis
- Myocarditis
- Aortic dissection in to coronary
- Left ventricular aneurysm
- Left ventricular pseudoaneurysm
- Early repolarization
- Left bundle branch block
- Left ventricular hypertrophy (LVH)
- Myocardial tumor
- Myocardial trauma
- RV pacing (appears as Left bundle branch block)
- Brugada syndrome
- Takotsubo cardiomyopathy
- AVR ST elevation
- Other thoracic
- Metabolic
- Drugs of abuse (eg, cocaine, crack, meth)
- Hyperkalemia (only leads V1 and V2)
- Hypothermia ("Osborn J waves")
- Medications
STEMI vs Pericarditis
| Disease | STEMI | Pericarditis |
| Pain | Constant | Varies with motion |
| Fever | No | Yes |
| ST changes | focal | Diffuse elevation |
| Reciprocal changes | Yes | No |
| Q waves | Yes | No |
| Pulmonary edema | Sometimes | No |
| Wall motion | Abnormal | Normal |
Evaluation
Workup
- ECG
- Use the J-point for measurement in 2 contiguous leads[10]
- J point is the junction between the end of the QRS complex and the beginning of the ST segment
- When possible, compare to old ECGs
- Repeating ECGs will increase sensitivity
- Use the J-point for measurement in 2 contiguous leads[10]
- CBC
- Chem 7
- Troponin
- PT/PTT
- Consider CXR
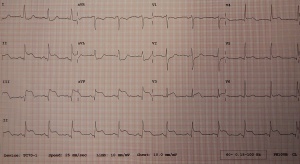
Diagnosis
Look for ST segment elevation, reciprocal ST depression, and hyperacute T waves
Classic STEMI
- Men
- Women
- ≥1.5 mm in V2-V3 and 1 mm (0.1mV) in all other leads[11]
Posterior STEMI
Up to 10% of STEMIs; usually associated with inferior MI
- ≥0.5 mm STE is diagnostic
- Look at V1-V3[12][13]
- Large R waves (posterior Q waves)
- STD
- Upright T waves
Post-arrest STEMI/NSTEMI
- Get immediate ECG after arrest
- STEMI: go to cath lab immediately (AHA/ACCF Class IB)[14]
- NSTEMI: go to cath within 2 hrs if VT/VF, intractable ischemic pain, ADCHF (AHA/ACC Class IA)[15]
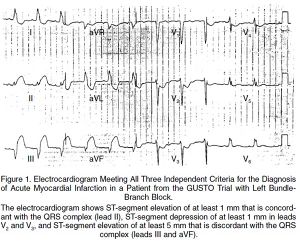
LBBB with Sgarbossa's criteria
- New LBBB alone is no longer STEMI criteria for cath lab as of 2013 per ACC/AHA guidelines[16]
- Hemodynamically unstable or new HF pts with new LBBB should be discussed with a cardiologist for PCI or fibrinolytics
- ≥3 points = 98% probability of STEMI[17]
- ST elevation ≥1 mm in a lead with upward (concordant) QRS complex - 5 points
- ST depression ≥1 mm in lead V1, V2, or V3 - 3 points
- ST elevation ≥5 mm in a lead with downward (discordant) QRS complex - 2 points
- Least specific of criteria, see Smith's modification
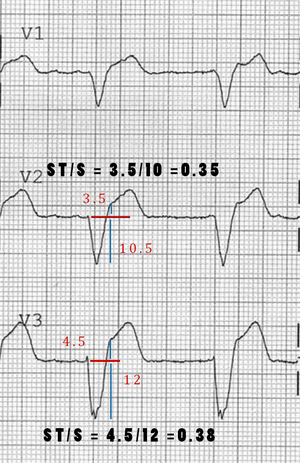
- Changes the 3rd rule of original Sgarbossa's Criteria to be ST depression OR elevation discordant with the QRS complex and with a magnitude of at least 25% of the QRS increases Sn from 52% to 91% at the expense of reducing Sp from 98% to 90%[18]
STEMI equivalents
- Pacemakers in AMI
- DeWinter T-waves[21]
- ST elevation in aVR[24]
- Reflects subendocardial ischemia in LV (L main vs multi vessal disease)[25]
- Look for STE >1-1.5 mm in aVR
- Can also bee seen in hemorrhage, type A Dissection, massive PE
- Aslanger criteria[26]
- Aslanger pattern is a reliable sign of acute inferior MI, indicating occlusion of the RCA or left circumflex artery
- Defined by a pattern consists of 3 criteria:
- ST elevation in leads III and aVRbut not in any other inferior lead
- ST depression in any of leads V4to V6but not in V2, AND
- ST in lead V1 higher than ST in lead V2.
- Leads III and aVR are contiguous in anatomical position; therefore, they should be recognized as contiguous leads on ECG.
Management
Thrombolytics vs PCI
Primary treatment is centered on PCI within 90 min (if available) or thrombolysis if treatment delay is greater than 120min.
- Percutaneous coronary intervention (preferred option)
- Goal: PCI should be attempted if the procedure can be started within 120 minutes (faster than 90 minutes is the goal, the faster the better)
- If the PCI cannot be commenced within 120 minutes then fibrinolysis should be given to suitable candidates
- Fibrinolytics
- Goal: if it is determined that PCI cannot be performed within 120 minutes then fibrinolytics should be given, and they should be given within 30 minutes
Adjunctive Therapies
- Aspirin 325mg PO or 600mg PR
- Nitroglycerin
- Should be avoided if concern for MI with RV extension or in patients with aortic stenosis
- Do not give if patient has used phosphodiesterase inhibitors (Viagra) in the past 24 hours
- O2
- Antiplatelet Options
- Clopidogrel
- See drug link for specific age, indication related dosages
- Generally, loading dose of 600 mg if PCI anticipated (otherwise give 300 mg)
- Ticagrelor
- May significantly reduce mortality as compared to clopidogrel[28]
- 180mg loading dose, followed by 90mg BID
- Ticagrelor offers no added benefit in STEMI when given pre-hospital vs. in-hospital (ambulance vs. cath lab)[29]
- Clopidogrel
- GPIIB/IIIa Inhibitors
- Abciximab, Eptifibatide
- Defer to cardiologist for administration
- Given right before PCI depending on specific institutional protocols
- Morphine
- Should be avoided if possible
- May interfere with the anti-platelet effect of P2Y12 receptor blockers (Aspirin)
- If pain is poorly controlled, consider 2-4mg IV with repeat dosing of 2-8mg ever 5-15 minutes
- Should be avoided if possible
- Beta blockers[30]
- Per AHA guidelines, IV beta blockers should not be given to patients with STEMI routinely, but may be considered for hypertension treatment barring contraindications as below:
- Low output state, signs of HF
- Increased risk of cardiogenic shock (age > 70, sinus tachycardia > 110 bpm or HR < 60 bpm, SBP < 120 mmHg)
- Increased time since onset of STEMI
- Relative contraindications -- asthma, COPD, PR interval > 0.24 sec, AV block
- PO beta blockers should be initiated within 24 hours of STEMI
- Per AHA guidelines, IV beta blockers should not be given to patients with STEMI routinely, but may be considered for hypertension treatment barring contraindications as below:
- ACE inhibitor or ARB[31][32]
- Give within 24 hours in stable patients, typically not given in ED
- Careful initial dosing, starting at 2.5 mg/day of lisinopril, increasing slowly up to 10 mg/day
- Statin in STEMI patients going to cath lab may have less 30-days MACEs and reinfarction post-PCI in SECURE-PCI trial in 2018[33]
- 80 mg atorvastatin immediately before cath lab and 24 hours after PCI
- Perform in conjunction with your cardiologist
- No cases of rhabdomyolysis or liver failure reported in the atorvastatin group
Anticoagulation
Heparin is required after thrombolysis to prevent re-thrombosis since all thrombolytics are short acting. Any patient receiving PCI requires heparinzation to prevent thrombosis during the procedure. There is minimal to no benefit for heparin in NSTEMI patients who are not receiving immediate PCI.[34]
- Heparin (UFH)
- Bolus 60U/kg (max: 4000U) followed by 12U/kg/h (max: 1000U/h)
- Titrate to PTT 1.5-2.5 x control
- LMWH
- <75yo with serum creatinine < 2.5mg/dL (men) or < 2.0mg/dL (women):
- 30mg IV bolus followed by 1mg/kg SC q12h
- ≥ 75yo
- 0.75mg/kg SC q12h
- CrCl < 30 mL/min
- 1mg/kg SC QD
- <75yo with serum creatinine < 2.5mg/dL (men) or < 2.0mg/dL (women):
- Fondaparinux
- creatinine < 3.0mg/dL:
- 2.5mg IV bolus then 2.5mg SC QD started 24hr after bolus
- Monitor anti-Xa levels
- creatinine < 3.0mg/dL:
- Bivalirudin
- 0.75mg/kg IV bolus followed by 1.75mg/kg/h
- CrCl < 30 mL/min
- 0.75mg/kg IV bolus followed by 1.0mg/kg/h
Special Scenarios
Cardiac Arrest and STEMI
- Consider therapeutic hypothermia cooling protocol for patients with documented cardiac arrest felt to be caused by lethal cardiac rhythm (e.g. ventricular fibrillation)
- Patients with cardiac arrest and ST elevation at any point, even if resolved, should still under go emergent coronary angiography[35]
Rescue PCI
- Failed reperfusion: consider if repeat ECG 90 minutes after infusion fails to show reduction of elevated ST segments by 50%
- Recurrent significant ST elevation following successful lysis
- Persistent hemodynamically unstable arrythmias, persistent ischemic symptoms, or worsened cardiogenic shock
- Even in those with successful reperfusion, its reasonable to do angiography within the index hospitalization, even within hours of thrombolytic therapy
Post-STEMI complications
- Cath site hematoma
- Heart failure or cardiogenic shock
- Hemodynamic support, consider norepinephrine or dobutamine
- Mechanical support e.g. balloon pump or Impella, usually placed in cath lab
- Ventricular tachycardia or ventricular fibrillation --> if sustained and > 48 hours after STEMI may need ICD
- Pericarditis, < 1 week from STEMI --> treat with high dose aspirin, avoid NSAIDs
- Bradycardia, if AV nodal involvement, rarely responds to atropine, likely will require pacing
- Free wall or ventricular septal rupture
- Dressler's syndrome, > 1 week post MI
Disposition
- Admit direct to cath lab
- If not at tertiary care center consider tPA depending transfer time and transfer to cardiac cath lab center
Calculators
See Also
- Acute Coronary Syndrome (Main)
- NSTEMI
- ACS - Anatomical Correlation
- Myocardial Infarction Complications
- ST segment elevation
- Sgarbossa's criteria
- STEMI equivalents
- STEMI mimics
External Links
- MDCalc - TIMI Risk Score for STEMI
- GRACE score - ACS risk model
- STEMI heart attack
- ACC-AHA guidelines for STEMI 2013
- EMCrit LBBB
References
- ↑ AHA ACA - NSTEMI ACS Guidelines 2014 View Online
- ↑ Thygesen, K et al. Fourth Universal Definition of Myocardial Infarction (2018). 2018 Nov 13;138(20):e618-e651.PMID: 30571511
- ↑ 3.0 3.1 Stub D et al. Air versus oxygen in ST-Segment-Elevation Myocardial Infarction. Circulation. 2015;121:2143-2150
- ↑ Ownbey M, Suffoletto B, Firsch A, et al. Prevalence and interventional outcomes of patients with resolution of ST-segment elevation between prehospital and in-hospital ECG. Prehosp Emerg Care. 2014. Apr-Jun;18(2):174-9
- ↑ Body R, Carley S, Wibberley C, et al. The value of symptoms and signs in the emergent diagnosis of acute coronary syndromes. Resuscitation. 2010;81(3):281–286. PMID: 20036454
- ↑ Panju AA, Hemmelgarn BR, Guyatt GH, et al. The rational clinical examination. Is this patient having a myocardial infarction? JAMA. 1998;280(14):1256–1263. PMID: 9786377
- ↑ Swap CJ, Nagurney JT. Value and limitations of chest pain history in the evaluation of patients with suspected acute coronary syndromes. JAMA. 2005;294(20):2623–2629. PMID: 16304077
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 Mehta LS, et al. Acute myocardial infarction in women: A scientific statement from the American Heart Association. Circulation. 2016; 133:916-947.
- ↑ Gimenez MR, et al. Sex-specific chest pain characteristics in the early diagnosis of acute myocardial infarction. JAMA Intern Med. 2014; 174(2):241-249.
- ↑ ACCF/AHA 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013 Jan 29;61(4):e78-140. PDF
- ↑ 11.0 11.1 11.2 Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD; Writing Group on behalf of the Joint ESC/ACCF/AHA/WHF Task Force for the Universal Definition of Myocardial Infarction.. Third universal definition of myocardial infarction. Glob Heart. 2012 Dec;7(4):275-95
- ↑ Ayer A, Terkelsen CJ. Difficult ECGs in STEMI: lessons learned from serial sampling of pre- and in-hospital ECGs. Journal of Electrocardiology 2014;47(4):448–58
- ↑ Wei EY, Hira RS, Huang HD, et al. Pitfalls in diagnosing ST elevation among patients with acute myocardial infarction. Journal of Electrocardiology 2013;46(6):653–9
- ↑ O’Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA Guideline for the Management of ST-Elevation Myocardial Infarction. JAC 2013;61(4):e78–e140.
- ↑ Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC Guideline for the Management of Patients with Non-ST-Elevation Acute Coronary Syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2014;64(24):e139–228.
- ↑ Am Heart J 2013;166:409-13
- ↑ Sgarbossa E. et al.. "Electrocardiographic diagnosis of evolving acute myocardial infarction in the presence of left bundle-branch block. GUSTO-1 (Global Utilization of Streptokinase and Tissue Plasminogen Activator for Occluded Coronary Arteries) Investigators". NEJM. 1996. 334(8):481-7
- ↑ Smith, S. et al. Diagnosis of ST-Elevation Myocardial Infarction in the Presence of Left Bundle Branch Block With the ST-Elevation to S-Wave Ratio in a Modified Sgarbossa Rule. 60(6). 766-776
- ↑ Sgarbossa EB, Pinski SL, Gates KB, et al. Early electrocardiographic diagnosis of acute myocardial infarction in the presence of ventricular paced rhythm. GUSTO-I investigators. Am J Cardiol. 1996;77(5):423–424.
- ↑ Maloy KR, Bhat R, Davis J, et al. Sgarbossa Criteria are Highly Specific for Acute Myocardial Infarction with Pacemakers.West J Emerg Med. 2010;11(4):354–357.
- ↑ Birnbaum I, Birnbaum Y. High-risk ECG patterns in ACS—need for guideline revision. J Electrocardiol. 2013;46(6):535–9.
- ↑ Verouden NJ, Koch KT, Peters RJ, et al. Persistent precordial “hyperacute” T-waves signify proximal left anterior descending artery occlusion.
- ↑ de Winter RJ, Verouden NJ, Wellens HJ, et al. A new ECG sign of proximal LAD occlusion. N Engl J Med. 2008 Nov 6;359(19):2071-3.
- ↑ Rokos IC, French WJ, Mattu A, et al. Appropriate cardiac cath lab activation: optimizing the electrocardiogram interpretation and clinical decision making for acute ST-elevation myocardial infarction. Am Heart J. 2010 Dec; 160(6):995-1003.
- ↑ Kosuge M, Uchida K, Imoto K, et al. An early and simple predictor of severe left main and/or three-vessel disease in patients with non-ST-segment elevation acute coronary syndrome. J Am Coll Cardiol. 2015 Jun 16;65(23):2570-1
- ↑ Zhang CH, Xu Z. Where Is the Culprit Lesion in the New Electrocardiogram Pattern?. JAMA Internal Medicine. March 7, 2022
- ↑ Stub et al. Air Versus Oxygen in ST-Segment Elevation Myocardial Infarction. Circulation. 2015 May 22.
- ↑ Wallentin et Al. Ticagrelor versus Clopidogrel in Patients with Acute Coronary Syndromes. N Engl J Med 2009; 361:1045-1057.
- ↑ Montalescot G et al. Prehospital ticagrelor in ST-segment elevation myocardial infarction. N Engl J Med 2014 Sep 1.
- ↑ Campbell-Schere DL, Green LA. ACC/AHA guideline update for the management of ST-segment elevation myocardial infarction. Am Fam Physician.2009 Jun 15;79(12):1080-6.
- ↑ ACE inhibitor use in patients with myocardial infarction. Summary of evidence from clinical trials. Latini R, Maggioni AP, Flather M, Sleight P, Tognoni G. Circulation. 1995;92(10):3132.
- ↑ Antman EM, et al. American College of Cardiology, American Heart Association Task Force on Practice Guidelines, Canadian Cardiovascular Society. Circulation. 2004;110(9):e82.
- ↑ Berwanger O, Santucci EV, de Barros e Silva PG, et al., on behalf of the SECURE-PCI Investigators. Effect of Loading Dose of Atorvastatin Prior to Planned Percutaneous Coronary Intervention on Major Adverse Cardiovascular Events in Acute Coronary Syndrome: The SECURE-PCI Randomized Clinical Trial. JAMA 2018;319:1331-40.
- ↑ Andrade-Castellanos, CA et al. Heparin versus placebo for non-ST elevation acute coronary syndromes. Cochrane Database Syst Rev. 2014 Jun 27;(6):CD003462. PMID: 24972265
- ↑ 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science PDF