Buprenorphine
Background
- Partial mu-opioid agonist and kappa-opioid antagonist used for opioid use disorder and chronic pain
- Has a ceiling effect for respiratory depression — at higher doses, respiratory depression plateaus, making it significantly safer than full agonists in overdose[1]
- ED-initiated buprenorphine increases 30-day treatment engagement ~5-fold compared to referral alone (D'Onofrio 2015 RCT)[2]
- Combined with naloxone in most sublingual/buccal formulations (Suboxone) to deter IV misuse — naloxone has no clinical effect when taken sublingually as intended
- No special waiver or certification is required to prescribe buprenorphine — the X-waiver (DATA 2000) was permanently eliminated by the Consolidated Appropriations Act of 2023[3]
Administration
- Type: Opioid — partial mu-agonist; used for OUD maintenance/induction and chronic pain
- Dosage Forms:
- Sublingual tablet: buprenorphine/naloxone 2 mg/0.5 mg, 4 mg/1 mg (generic), 8 mg/2 mg, 12 mg/3 mg; buprenorphine monoproduct 2 mg, 8 mg
- Sublingual/buccal film: buprenorphine/naloxone 2 mg/0.5 mg, 4 mg/1 mg, 8 mg/2 mg, 12 mg/3 mg (Suboxone); buprenorphine buccal film 75, 150, 300, 450, 600, 750, 900 mcg (Belbuca — for pain)
- Extended-release subcutaneous injection: Sublocade 100 mg, 300 mg (monthly); Brixadi 8, 16, 24, 32 mg (weekly) and 64, 96, 128 mg (monthly)
- Transdermal patch: 5, 7.5, 10, 15, 20 mcg/hr (Butrans — for pain)
- Injectable solution: 0.3 mg/mL (Buprenex — for acute pain, available IV/IM)
- Routes of Administration: sublingual, buccal, subcutaneous, transdermal, IV, IM
- Common Trade Names: Suboxone (buprenorphine/naloxone), Subutex (buprenorphine mono), Sublocade (extended-release monthly), Brixadi (extended-release weekly/monthly), Belbuca (buccal film for pain), Butrans (transdermal patch for pain), Buprenex (injectable)
Adult Dosing
ED-Initiated Buprenorphine
There are three induction strategies. Choice depends on withdrawal status and clinical scenario.[4][1]
Traditional Induction (patient in moderate-severe withdrawal, COWS ≥8-12)
- For Clinical Opioid Withdrawal Scale (COWS) ≥8: give 4 to 8mg of Buprenorphine, observe 30 to 45min
- Redose if COWS remains ≥8. Then discharge home with 16 mg a day to bridge until follow-up (an X-waiver is no longer required to prescribe buprenorphine).
- For Clinical Opioid Withdrawal Scale (COWS) 0-7: Consider observing the patient until their COWS score is >8 for the standard buprenorphine induction.
- If sublingual tablets/films unavailable then intravenous/intramuscular formulation (dose 0.3-0.9 mg every 6-12 hours) has been used for opioid withdrawal in the ED[9] and hospitalized[10] patients.
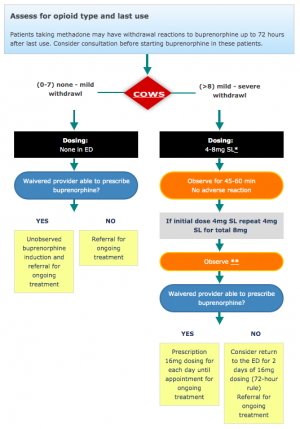
NIH National Institute on Drug Abuse ED Buprenorphine algorithm[11]
High-Dose ("Macro") Induction
- For patients in moderate-severe withdrawal, especially those using fentanyl/high-potency synthetic opioids
- Day 1: Buprenorphine/naloxone 8 mg SL; may repeat 8 mg in 1-2 hours (total up to 16-32 mg on day 1)
- Buprenorphine has been shown to be safe at doses up to 32-64 mg without respiratory depression[1]
- If precipitated withdrawal occurs with the initial dose, give more buprenorphine (not less) to saturate remaining unoccupied receptors[1]
- Discharge dose: 16 mg/day (or the total daily dose that stabilized the patient in the ED), prescribed as buprenorphine/naloxone with outpatient follow-up
Low-Dose ("Micro") Induction
- For patients not yet in withdrawal or who cannot tolerate withdrawal (e.g., patients on full-agonist opioids, patients using fentanyl who cannot wait for COWS ≥8)
- Start with very low doses of buprenorphine (e.g., 0.5-1 mg SL) and gradually increase over 3-7 days while the full agonist is slowly tapered
- Avoids precipitated withdrawal by allowing gradual receptor binding
- Typically not completed in a single ED visit — requires either observation unit admission or close outpatient follow-up[4]
Precipitated Withdrawal Management
- Occurs when buprenorphine is given while a full agonist is still occupying mu receptors (more common with fentanyl due to lipophilicity and depot effect)
- Treat with more buprenorphine — increasing the dose is first-line to overcome competition for receptor binding[1]
- Adjunctive symptom management:
- Ondansetron for nausea/vomiting
- Loperamide for diarrhea
- Clonidine 0.1-0.2 mg for autonomic symptoms
- NSAIDs or acetaminophen for myalgias
- IV fluids for dehydration
Acute Pain (non-OUD)
- IV/IM: 0.3 mg q6-8h PRN (parenteral formulation)
- Transdermal (Butrans): 5-20 mcg/hr patch, changed weekly
- Buccal film (Belbuca): Starting dose 75 mcg q12h, titrate as needed
Pediatric Dosing
- OUD: Generally managed by addiction medicine/pediatric specialists; weight-based dosing for adolescents ≥16 years follows adult guidelines
- Acute pain (parenteral): 2-12 years: 2-6 mcg/kg q4-6h PRN
Special Populations
Pregnancy
- Buprenorphine is recommended in pregnancy for OUD — associated with reduced neonatal abstinence syndrome (NAS) severity and shorter NICU stays compared to methadone[12]
- Use buprenorphine monoproduct (without naloxone) in pregnancy — although buprenorphine/naloxone is increasingly accepted and may be used if the monoproduct is unavailable[13]
- Do NOT stop buprenorphine in pregnancy — withdrawal poses greater risk to fetus than continued treatment
Lactation
- Buprenorphine is considered compatible with breastfeeding — minimal transfer to breast milk, low oral bioavailability in the infant[13]
- Benefits of breastfeeding generally outweigh risks in stable patients on buprenorphine maintenance
- Monitor infant for sedation and adequate weight gain
Renal Dosing
- No dose adjustment required (primarily hepatically metabolized)[1]
Hepatic Dosing
- Use with caution in moderate hepatic impairment; reduce dose
- Avoid or do not use in severe hepatic impairment
- Extended-release formulations (Sublocade, Brixadi) are contraindicated in moderate-severe hepatic impairment (cannot rapidly adjust dose once injected)
- Obtain LFTs if clinical concern, but LFTs are not required before initiating buprenorphine in the ED[1]
Contraindications
- Allergy to buprenorphine or naloxone
- Severe respiratory depression (current; note: ceiling effect makes this very rare with buprenorphine alone)
- Severe hepatic impairment (for extended-release formulations)
- Not contraindicated in patients on benzodiazepines — co-prescribing carries risk but withholding buprenorphine carries greater mortality risk from untreated OUD[4]
Drug Interactions
- Benzodiazepines and other CNS depressants: Additive respiratory depression risk, but this is NOT a contraindication to buprenorphine initiation — the risk of death from untreated OUD exceeds the risk of co-prescribing[4]
- CYP3A4 inhibitors (ketoconazole, ritonavir, erythromycin): May increase buprenorphine levels — monitor for sedation
- CYP3A4 inducers (rifampin, carbamazepine, phenytoin): May decrease buprenorphine levels — may require dose increase
- Full mu-agonist opioids: Buprenorphine may precipitate withdrawal if given while full agonist is occupying receptors; full agonists may have diminished effect in patients on buprenorphine maintenance
- Naloxone: Higher doses of naloxone (standard overdose doses) may precipitate withdrawal in buprenorphine-dependent patients, though buprenorphine's high receptor affinity makes it relatively naloxone-resistant
Adverse Reactions
Serious
- Respiratory depression (rare due to ceiling effect; risk increased with concurrent CNS depressants or IV misuse)
- Precipitated opioid withdrawal (if given before full agonist has cleared)
- Hepatotoxicity (elevated transaminases; more common with pre-existing hepatic disease or hepatitis)
- QT prolongation (mild; less clinically significant than with methadone)
- Adrenal insufficiency (rare, with chronic use)
- Neonatal withdrawal syndrome (in infants born to mothers on buprenorphine)
- Anaphylaxis/angioedema (rare)
Common
- Constipation
- Nausea, vomiting
- Headache
- Insomnia
- Diaphoresis
- Peripheral edema
- Dizziness, somnolence
- Oral hypoesthesia (sublingual/buccal formulations)
- Injection site reactions (extended-release formulations)
Pharmacology
- Half-life: 24-42 hours (sublingual); 43-60 days (Sublocade depot)
- Metabolism: Hepatic via CYP3A4 to active metabolite norbuprenorphine; also undergoes glucuronidation
- Excretion: Primarily fecal (~70%); renal (~30% as metabolites)
- Protein binding: ~96%
- Ceiling effect for respiratory depression occurs at approximately 16-32 mg sublingual[1]
Mechanism of Action
- Partial mu-opioid receptor agonist: Produces submaximal activation — provides analgesia and prevents withdrawal but with limited euphoria and a ceiling effect on respiratory depression
- Kappa-opioid receptor antagonist: May contribute to antidepressant and anti-dysphoric effects
- Very high receptor binding affinity: Displaces full agonists from mu receptors (causes precipitated withdrawal if administered too early) and blocks effects of subsequently administered full agonists
Comments
- ED-initiated buprenorphine is the standard of care — supported by ACEP policy statement, ASAM guidelines, and landmark RCT data showing reduced opioid use, increased treatment engagement, and reduced mortality[2][14]
- Prescribe a bridge supply at discharge: At minimum, prescribe enough buprenorphine/naloxone to last until the outpatient follow-up appointment (typically 7-14 day supply). A common discharge dose is 16 mg/day[1]
- Buprenorphine and buprenorphine/naloxone are clinically interchangeable. Buprenorphine/naloxone is now generic in the US
- Patients presenting on extended-release buprenorphine (Sublocade, Brixadi) who have breakthrough withdrawal may need supplemental sublingual buprenorphine — consult with the patient's prescriber
- For patients with OUD in the ED who decline buprenorphine, still treat withdrawal symptoms, provide harm reduction resources (naloxone kit, fentanyl test strips), and offer a prescription or referral for a future visit
Indications by Condition
The following table is automatically generated from disease/condition pages across WikEM.
| Indication | Dose | Context | Route | Population |
|---|---|---|---|---|
| Neonatal abstinence syndrome | 5mcg/kg q8hr initially (max 20mcg/kg q8hr) | Alternative to morphine; may reduce hospital stay | Sublingual | Pediatric |
| Opioid toxicity | 4 mg SL initial; may give additional 4 mg SL after 1-2 hours (total 8-16 mg on day 1) | ED-initiated buprenorphine for OUD | SL | Adult |
See Also
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 Hughes T, Nasser N, Mitra A. Overview of best practices for buprenorphine initiation in the emergency department. Int J Emerg Med. 2024;17:23. doi:10.1186/s12245-024-00593-6
- ↑ 2.0 2.1 D'Onofrio G, O'Connor PG, Pantalon MV, et al. Emergency department-initiated buprenorphine/naloxone treatment for opioid dependence: a randomized clinical trial. JAMA. 2015;313(16):1636-1644.
- ↑ Consolidated Appropriations Act, 2023, Pub. L. No. 117-328, § 1262, 136 Stat. 4459 (2022).
- ↑ 4.0 4.1 4.2 4.3 Weimer MB, Herring AA, Kawasaki SS, Meyer M, Kleykamp BA, Ramsey KS. ASAM Clinical Considerations: Buprenorphine Treatment of Opioid Use Disorder for Individuals Using High-potency Synthetic Opioids. J Addict Med. 2023;17(6):632-639. doi:10.1097/ADM.0000000000001202
- ↑ A Guide for Patients Beginning Buprenorphine Treatment at Home https://medicine.yale.edu/edbup/quickstart/Home_Buprenorphine_Initiation_338574_42801_v1.pdf
- ↑ A Patient’s Guide to Starting Buprenorphine at Home from ASAM https://www.asam.org/docs/default-source/education-docs/unobserved-home-induction-patient-guide.pdf
- ↑ Buprenorphine Home Induction Apple App Store https://apps.apple.com/us/app/buprenorphine-home-induction/id1449302173
- ↑ Starting Buprenorphine from Google Play Store https://play.google.com/store/apps/details?id=com.amstonstudio.bup&hl=en_US&gl=US
- ↑ Berg ML, et. al. Evaluation of the use of buprenorphine for opioid withdrawal in an emergency department. Drug Alcohol Depend. 2007 Jan 12;86(2-3):239-44. https://doi.org/10.1016/j.drugalcdep.2006.06.014. Epub 2006 Aug 22. PMID: 16930865.
- ↑ Welsh CJ, Suman M, Cohen A, et al. The use of intravenous buprenorphine for the treatment of opioid withdrawal in medically ill hospitalized patients. Am J Addict. 2002;11(2):135-40 https://doi.org/10.1080/105500490290087901
- ↑ https://d14rmgtrwzf5a.cloudfront.net/sites/default/files/algorithm.pdf
- ↑ Jones HE, Kaltenbach K, Heil SH, et al. Neonatal abstinence syndrome after methadone or buprenorphine exposure. N Engl J Med. 2010;363(24):2320-2331.
- ↑ 13.0 13.1 ACOG Committee Opinion No. 711: Opioid Use and Opioid Use Disorder in Pregnancy. Obstet Gynecol. 2017;130(2):e81-e94.
- ↑ Armour R, et al. Initiation of buprenorphine in the emergency department or emergency out-of-hospital setting: A mixed-methods systematic review. Am J Emerg Med. 2025;88:12-22.


